Materials Required:
- Spectrophotometer
- Cuvette
- Blank solution
Reagents:
- Cobalt (II) chloride
- Hexaaquacobalt (II) ion
- Ferrocene
- Crystal violet
- Rose bengal
- Coumarin
Procedure:
Determination of Molar Absorption Coefficient:
- Select a blank cuvette and place it in the spectrophotometer. Close the lid.
- Click on 0 ABS 100%T button, the instrument now reads 0.00000 A.
- Choose a solution with known concentration and measure the absorbance between the wavelengths 350 nm to 700 nm.
- Record the wavelength at the maximum absorbance value.
- Calculate the value of molar absorption coefficient , using the equation
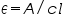 .
.
Determination of Unknown Concentration:
- Set the wavelength to the value corresponding to maximum absorbance (recorded above).
- Place the cuvette with same solution but at an unknown concentration.
- Read the absorbance for this wavelength.
- Calculate the concentration with the help of the equation, molarity
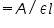
- Enter the calculated concentration value in the given box. (Note : Should enter the value correct to four decimal places)
- Repeat the same procedure for a second solution.
Observations and Calculations:
Concentration of Solution =.............................M
of Solution =.............................M
Absorbance of known conc. solution =...............................A
of known conc. solution =...............................A
Cuvette length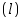 =.............................cm
=.............................cm
Wavelength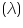 at maximum absorbance value =..............................nm
at maximum absorbance value =..............................nm
Molar Absorption Coefficient,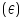 =............................. M−1 cm−1
=............................. M−1 cm−1
Absorbance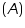 of given solution =..............................A
of given solution =..............................A
Therefore, concentration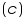 =.............................M
=.............................M
Result:
Concentration of the given solution =...............................M
Points to Remember while Performing the Experiment in a Real Laboratory:
- Always wear lab coat and gloves when you are in the lab. When you enter the lab, switch on the exhaust fan and make sure that all the chemicals and reagents required for the experiment are available. If they are not available, prepare the reagents using the components for reagent preparation.
- Make sure to clean all your working apparatus with chromic acid and distilled water and ensure that all the apparatus are free from water droplets while performing the experiment.
- Make sure to calibrate the electronic weigh balance before taking the measurements.
- Ensure that the spectrophotometer is working properly.
- Ensure that you are handling the cuvette with tissue paper .Never touch it with your hand.
- Wipe the cuvette with tissue paper before placing the spectrophotometer.
- Clean all glassware with soap and distilled water. Once the experiment is completed recap the reagent bottles. Switch off the light and exhaust fan before leaving the lab.
- Discard the used gloves in a waste bin.




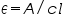 .
.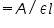
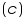 of Solution
of Solution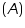 of known conc. solution
of known conc. solution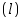
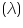 at maximum absorbance value
at maximum absorbance value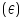 =.............................
=.............................