Materials Required:
- Polarographic Instrument.
- Beaker.
- 10 mL Pipette.
- Wash bottle.
- 100 mL volumetric flask.
Reagents:
- 1, 2, 3, 4 and 5 mM Cadmium standards.
- 2 M KCl solution.
- 0.2% gelatin solution.
- Distilled water.
Procedure:
- Select the concentration from the list.
- Click “Load Cadmium Sample” button.
- Select “Scan Analysis”.
- Click “Plot Graph”.
- Select the unknown concentration from the concentration list.
- Repeat the steps 2, 3 and 4.
- Enter the concentration values on the worksheet.
- Plot calibration curve by clicking the “plot” button on the worksheet.
- Calculate the unknown concentration value from the calibration curve.
Observations and Calculations:
| No |
Concentration (mM) |
Diffusion current (id) 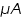
|
| 1 |
|
|
| 2 |
|
|
| 3 |
|
|
| 4 |
|
|
| 5 |
|
|
Plot concentration Vs difussion current.
id of unknown concentration =............................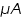 .
.
Therefore concentration of the given solution =......................................mM.
Result:
Unknown concentration of the given solution =.......................mM.
Points to Remember while Performing the Experiment in a Real Laboratory:
- Always wear lab coat and gloves when you are in the lab. When you enter the lab, switch on the exhaust fan and make sure that all the chemicals and reagents required for the experiment are available. If it is not available, prepare the reagents using the components for reagent preparation.
- Make sure to clean all your working apparatus with chromic acid and distilled water and ensure that all the apparatus are free from water droplets while performing the experiment.
- Make sure to calibrate the electronic weigh balance before taking the measurements.
- Ensure that the spectrophotometer is working properly.
- Ensure that you are handling the cuvette with tissue paper. Never touch it with your hand.
- Wipe the cuvette with tissue paper before placing the spectrophotometer.
- Clean all glass wares with soap and distilled water. Once the experiment completed recap the reagent bottles. Switch off the light and exhaust fan before leaving the lab.
- Discard the used gloves in a waste bin.