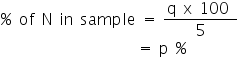Procedure:
-
Determination of Available Nitrogen Content in Soil:
- Weigh 50 g of processed soil sample in 500 mL Kjeldahl flask.
- Add 1 g CuSO4, 10 g K2SO4 and 30 mL Con. H2SO4.
- Shake the contents of the flask until through mixing and allowing to stand for at least 30 minutes with frequent shaking or until complete solution results.
- Digest the content until greenish colour appears. K2SO4 raises the boiling point of the acid. So that the loss of acid volatile solution is prevented. CuSO4 5H2O is digestion accelerator which catalyses the speed of digestion process.
- The reagents sometimes contain impurities so run a blank with the same quantities of reagents and subtract the blank value from the value of the soil digest.
- Digestion is effected on the Kjeldahl digestion rack with low flame for the first 10 – 30 min until the frothing stops and then gradually more strongly until the sample is completely charred. The heat is gradually raised until the acid reaches approximately one third the way up the digestion-flask. The flame is not allowed to touch the flask above the part occurred by the liquid. Excessive boiling may cause volatilization of the acid before the organic matter is oxidized.
- Cool the content and dilute to about 100 mL with distilled water. Swirl the flask for about 2 minutes and transfer the fluid part to a 1000 mL distillation flask.
- Wash the residue left in the Kjeldahl flask with 4 or 5 lots of 50 – 60 mL distilled water, decanting the washings into the distillation flask.
- Add a few, glass bead to prevent bumping.
- Fit the flask with two neck joints to one neck dropping funnel is connected for adding 40 % NaOH while to the other neck Kjeldahl trap, which is used to trap the NaOH coming with the distillate. The trap is connected to the condenser with a delivery tube which dips into 50 mL of 0.1 N HCl contained in a conical flask, with one or two drops of methyl red indicator.
- Add about 125 mL (or 100 ml if bumping is a problem) of 40 % NaOH solution till the content are alkaline in reaction (about 5 times the volume of Con. H2SO4 used during the digestion). Heat the RB flask.
- Allow the ammonia formed to be absorbed in standard HCl. Wash down the end of the tube. 150 mL distilled water is added to the conical flask. When no more ammonia is received (test with a red litmus paper turning blue) stop the distillation.
- Titrate the excess of the acid with 0.1 N NaOH solution till the pink colour changes to yellow.
- From the titre value calculate the multi equivalence of the acid participating in the process of ammonia absorbing during digestion.
a) Calculation:
i. Blank:
Volume of HCl taken for blank = a mL
Volume of NaOH used = b mL
Volume of HCl consumed by liberated NH3 present in blank = a – b = z mL
ii. Sample:
Volume of HCl taken for sample = v mL
Volume of NaOH used = u mL
Volume of HCl consumed by liberated NH3 present in sample = v – u = w mL
Volume of HCl consumed for NH3 liberated by sample only = w – z = y mL
1000 mL 1N HCl = 1000 mL 1 N NH3 = 17 g NH3 = 14 g N
1 mL 1N HCl = 1 mL 1 N NH3 = 0.014 g N
1 mL 0.1 N HCl = 1 mL 0.1 N NH3 = 0.0014 g N
Weight of Nitrogen in 5 g of Sample = y x 0.0014 g N = q g N
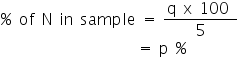
Points to Remember while Performing the Experiment in a Real Laboratory:
- Always wear lab coat and gloves when you are in the lab. When you enter the lab, switch on the exhaust fan and make sure that all the chemicals and reagents required for the experiment are available. If it is not available, prepare the reagents using the components for reagent preparation.
- Properly adjust the flame of the Bunsen burner. The proper flame is a small blue cone; it is not a large plume, nor is it orange.
- Make sure to clean all your working apparatus with chromic acid and distilled water and ensure that all the apparatus are free from water droplets while performing the experiment.
- Make sure to calibrate the electronic weigh balance before taking the measurements.
- Ensure that the desiccator has sufficient amount of desiccant; Silica gel
- Use chromic acid to clean the crucible, then heat it and make sure to cool it and before placing in the desiccators. Ensure that you are handling the crucible, with cleaned tongs or with tissue paper .Never touch it with your hand.
- Switch on the oven and adjust the temperature to 1300 C. Make sure to use a cotton glove while working with a hot air oven.
- Make sure to clean the Kipp's apparatus tube with water and ensure that it has sufficient solid material; iron sulfinide and acid, H2SO4 for producing H2S gas.
- Clean all glass wares with soap and distilled water. Once the experiment completed recap the reagent bottles. Switch off the light, exhaust fan, hot air oven and Gas cylinder before leaving the lab.
- Discard the used gloves in a waste bin.